What Are The Properties Between Chenodeoxycholic Acid And Ursodeoxycholic Acid?
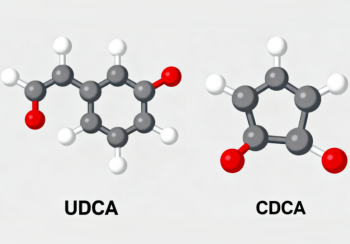
Structure:
Both molecules are C24 bile acids with two hydroxyl groups (a 3α-hydroxyl common to many bile acids, and a 7-hydroxyl). The orientation (α or β) of the 7-hydroxyl distinguishes them: CDCA is 3α,7α-dihydroxy-5β-cholan-24-oic acid (7α-OH), and UDCA powder is the 7β-epimer (3α,7β-dihydroxy-5β-cholan-24-oic acid). This epimerization at C-7 changes crystal packing, solubility behavior and biological interactions.

Hydrophobicity/hydrophilicity:
CDCA is more hydrophobic (less water-friendly) than UDCA. Ursodeoxycholic Acid powder is relatively hydrophilic and is considered a "hydrophilic" bile acid compared with CDCA and other more hydrophobic bile acids (like deoxycholic acid and lithocholic acid). This difference explains much of their divergent cellular toxicity and protective capacity: hydrophobic bile acids are detergent-like and can damage cell membranes and organelles; hydrophilic bile acids are less damaging and can even protect cells by displacing more hydrophobic bile acids from bile-acid pools.
Physical consequences:
The shift from α- to β-hydroxyl changes the molecule's three-dimensional surface and hence binding to proteins and receptors, as well as the propensity to form micelles, interact with membranes, and be modified by gut bacteria. Crystal-structure studies and comparative physical chemistry papers document these differences.
How Do Chenodeoxycholic Acid And Ursodeoxycholic Acid Recycle Bile Acids?

Natural occurrence:
In humans, primary bile acids synthesized in the liver from cholesterol are cholic acid and chenodeoxycholic acid (CDCA). Ursodeoxycholic acid mechanism is present in small amounts in humans but is more abundant in some other mammals (e.g., bears). Gut microbiota can epimerize and dehydroxylate bile acids. Bacterial 7α-dehydroxylation and epimerization pathways can convert CDCA into other products and can participate in interconversion between CDCA and UDCA bulk powder depending on microbial enzymes present.
Conjugation and recirculation:
Both CDCA and UDCA are conjugated in the liver (glycine or taurine conjugates) before secretion into bile, are released into the intestine where bacteria act on them, and are largely reabsorbed in the terminal ileum. Differences in how gut bacteria act on them contribute to differences in pool composition and downstream effects on lipid metabolism.

What Are The Different Uses Between Chenodeoxycholic Acid And Ursodeoxycholic Acid?
Below are the principal clinical indications and how CDCA and UDCA compare.
Cholesterol gallstones
Both can dissolve small, radiolucent cholesterol gallstones by reducing cholesterol saturation in bile and slowly shrinking stones. Historically, both chenodeoxycholic acid (chenodiol) and ursodeoxycholic acid (urso/ursodiol) were used for nonsurgical dissolution of cholesterol gallstones.
Comparative outcomes and side effects:
Clinical studies from the 1970s–1980s showed that UDCA generally dissolves stones faster and with fewer adverse effects than CDCA; CDCA tended to be more effective at higher doses for small stones but caused more gastrointestinal and systemic side effects (e.g., diarrhea, abnormal liver tests in some cases). Over time, Ursodeoxycholic Acid bulk powder became preferred for gallstone dissolution because of its safer profile and comparable long-term efficacy.
Cholestatic liver diseases
- UDCA (ursodiol)
Ursodeoxycholic Acid is the established first-line medical therapy for primary biliary cholangitis (PBC). Long-term UDCA improves biochemical cholestatic markers (alkaline phosphatase, bilirubin) and is associated with improved transplant-free survival when started early; it is also used in other cholestatic settings (selected primary sclerosing cholangitis patients, intrahepatic cholestasis of pregnancy, cholestasis from parenteral nutrition) with variable evidence. UDCA's choleretic and cytoprotective effects make it the mainstay for these disorders.
- CDCA in cholestasis:
Because CDCA is relatively hydrophobic and can be hepatotoxic at higher concentrations, it is not used as a general therapy for cholestatic liver diseases and is not the standard for PBC. CDCA's FXR activity might theoretically be useful in some metabolic situations, but Ursodeoxycholic Acid UDCA remains the clinically established therapy for cholestatic disorders.
Inborn errors of bile-acid synthesis and pediatric enzyme deficiencies
- CDCA therapy for certain bile-acid synthesis defects:
A key use of CDCA as a therapeutic agent is in specific inborn errors of bile-acid synthesis (for example, some single-enzyme deficiencies) and in cerebrotendinous xanthomatosis (CTX). In CTX or certain synthesis defects, CDCA administration provides feedback inhibition of abnormal bile-acid synthesis pathways and can reduce the production of toxic intermediates; CDCA replacement can correct metabolic imbalance. Thus CDCA has a role in genetic bile-acid disorders that Ursodeoxycholic Acid UDCA does not fulfill as effectively.
Lipid metabolism, metabolic effects, and cardiovascular implications
- FXR-dependent lipid effects with CDCA:
CDCA's potent FXR activation can lower bile-acid synthesis and alter lipoprotein handling. FXR activation can reduce hepatic de novo bile-acid synthesis and affect LDL/PCSK9 pathways; these metabolic effects are being researched therapeutically (and synthetic FXR agonists have been developed). Thus CDCA has a stronger and more direct influence on bile-acid–sensitive metabolic circuits than UDCA does.
- UDCA and lipids:
Ursodeoxycholic acid mechanism of action on systemic lipid profiles is weaker and less predictable because its major actions are local (bile composition, hepatocyte protection) rather than potent transcriptional reprogramming through FXR.
What is the Safety Between Chenodeoxycholic Acid And Ursodeoxycholic Acid?

CDCA:
Because CDCA is more hydrophobic and a stronger FXR agonist, it can be more likely to produce side effects such as diarrhea, elevated liver enzymes, and-rarely-worse hepatotoxicity in some cholestatic contexts. Historically, higher rates of gastrointestinal adverse effects limited its long-term tolerability relative to UDCA in gallstone-dissolution protocols. CDCA is used carefully and in specific metabolic disorders where its benefits outweigh risks.
UDCA:
Is ursodeoxycholic acid safe? Generally well tolerated. Common side effects are mild gastrointestinal complaints (e.g., diarrhea, bloating) in a minority of patients. Because Ursodeoxycholic Acid displaces more hydrophobic bile acids, it usually reduces cytotoxicity rather than increasing it. In cholestatic disease long-term safety has been extensively documented. Rarely, ursodeoxycholic acid (UDCA) may worsen outcomes in some conditions (historical cautionary findings in certain trials), so clinical context matters.

What is the Dosage Between Chenodeoxycholic Acid And Ursodeoxycholic Acid?
Administration:
Both are given orally and are absorbed, conjugated in the liver, secreted in bile, and largely reabsorbed (enterohepatic circulation). Conjugation (with glycine or taurine) influences water solubility and biliary secretion.
Typical dosing:
Ursodeoxycholic Acid dosing for PBC is commonly around 13–15 mg/kg/day in divided doses (clinical practice guidelines). For gallstone dissolution, smaller doses (e.g., 8–10 mg/kg/day) have been used historically.
CDCA (chenodiol) dosing when used for gallstone disease historically involved a range (e.g., 10–15 mg/kg/day) but may cause more adverse effects at higher regimens. Exact dosing depends on the indication and individual patient factors; specialist guidance is required for CDCA used in rare metabolic disorders. (Note: always follow local guidelines and specialist prescriptions.)
Conclusion:
In summary, chenodeoxycholic acid and ursodeoxycholic acid, despite being epimers differing only in the orientation of a single hydroxyl group, are molecular doppelgangers with opposing natures. CDCA, a primary human bile acid, is hydrophobic, membrane-disruptive, and cytotoxic. Its therapeutic role is confined to the specific metabolic defect of Cerebrotendinous Xanthomatosis, where it acts to restore a crucial metabolic feedback loop. In contrast, pure UDCA, a hydrophilic and cytoprotective molecule, has emerged as a versatile and safe hepatoprotective agent. Its ability to detoxify the bile acid pool, stimulate bicarbonate-rich bile flow, and protect cells from apoptosis underpins its success in treating a spectrum of cholestatic liver diseases, from PBC to ICP. CDCA and Ursodeoxycholic Acid affect the molecular stereochemistry on biological function. It underscores a fundamental principle in pharmacology: a minor structural change can be the difference between a toxin and a therapy.
The global demand for high-purity ursodeoxycholic acid UDCA for pharmaceutical use is met by specialized manufacturers, and Guanjie Biotech is a bulk ursodeoxycholic acid manufacturer playing a critical role in this supply chain, ensuring that this vital medicine reaches patients worldwide. As one of the professional ursodeoxycholic acid manufacturers, we can provide high-quality ursodeoxycholic acid. If you need our ursodeoxycholic acid powder, welcome to enquire with us at info@gybiotech.com.
References
[1] Hofmann, A. F., & Hagey, L. R. (2014). Key discoveries in bile acid chemistry and biology and their clinical applications: history of the last eight decades. Journal of Lipid Research, *55*(8), 1553–1595.2.
[2] Poupon, R. (2012). Ursodeoxycholic acid and bile-acid mimetics as therapeutic agents for cholestatic liver diseases: mechanisms of action and clinical applications. Seminars in Liver Disease, *32*(1), 066–073.
[3] Paumgartner G, Beuers U. Ursodeoxycholic acid in cholestatic liver disease: mechanisms of action and therapeutic efficacy. Hepatology. 2004.
[4] Roda E, Fromm H, et al. Ursodeoxycholic acid vs. chenodeoxycholic acid in gallstone dissolution: comparative study. Hepatology / Gastroenterology literature (1982).
[5] Trauner M, Boyer JL. Mechanisms of action and therapeutic efficacy of ursodeoxycholic acid in cholestatic liver disease. 1999 review.
[6] Guo C, et al. Farnesoid X receptor agonists: roles of CDCA and synthetic agonists (e.g., obeticholic acid). Journal reviews (2018).
[7] DrugBank / PubChem entries for chenodeoxycholic acid and ursodeoxycholic acid - identification, structure and clinical use summaries.
[8] Lazaridis KN, et al. Ursodeoxycholic acid mechanisms and clinical implications. Journal of Hepatology review (2001).
[9] Van Hooff MC, et al. Treatment in primary biliary cholangitis: beyond UDCA - recent reviews and guidelines (2024). European Journal reviews.
[10] Hirano S. Bacterial epimerization and dehydroxylation of bile acids. Microbial metabolism literature.










