Nervonic acid NA is a long-chain monounsaturated fatty acid. It belongs to the category of Very Long-Chain Fatty Acids. As a structurally specialized member of the fatty acid family, nervonic acid's synthesis depends on other fatty acid precursors, and it plays an irreplaceable role in the myelin sheath structure of the central nervous system. What is the relationship between nervonic acid and fatty acids?

What Is The Relationship Between Nervonic Acid And Fatty Acids?
Positioning of Nervonic Acid in the Fatty Acid
Fatty acids are organic compounds composed of hydrocarbon chains and terminal carboxyl groups. Based on carbon chain length, fatty acids can be classified into short-chain fatty acids (C2-C6), medium-chain fatty acids (C8-C14), long-chain fatty acids (C16-C18), and very long-chain fatty acids (C20 and above). Nervonic acid has 24 carbon atoms, classifying it as a typical very long-chain fatty acid.
Based on unsaturation, fatty acids are classified into saturated fatty acids (no double bonds), monounsaturated fatty acids (containing one double bond), and polyunsaturated fatty acids (containing two or more double bonds). Nervonic acid contains a double bond between the 15th and 16th carbon atoms in its carbon chain. Therefore, it is classified as a monounsaturated fatty acid.
Based on the position of the double bond, monounsaturated fatty acids are further divided into the ω-9 series (double bond located between the 9th and 10th carbons counting from the methyl end) and the ω-7 series, etc. The double bond position of nervonic acid is Δ15, which is between the 9th and 10th carbons counting from the methyl end; therefore, it belongs to the ω-9 monounsaturated fatty acid category. Other members of this family include oleic acid and erucic acid.
The above classification relationship can be summarized as: nervonic acid → long-chain fatty acids → monounsaturated fatty acids → ω-9 fatty acids.
Biosynthetic Pathway of Nervonic Acid
The synthesis of nervonic acid in organisms does not occur independently but depends on the carbon chain elongation and desaturation reactions of long-chain fatty acids. The synthetic pathway consists of two stages.
Stage 1: Conversion of Oleic Acid to Erucic Acid
Oleic acid (18:1Δ9) is the shortest-chain member of the ω-9 fatty acid family and serves as the starting point for nervonic acid synthesis. Catalyzed by an endoplasmic reticulum membrane-bound fatty acid elongase complex, oleic acid undergoes two consecutive carbon chain elongation reactions:
Oleic acid (18:1Δ9) is elongated by elongase to generate eicosenoic acid (20:1Δ11).
Eicosenoic acid is further elongated to generate erucic acid (22:1Δ13).
Stage 2: Elongation of Erucic Acid to Nervonic Acid
Erucic acid (22:1Δ13) is catalyzed by a fatty acid elongase complex, which adds two carbon atoms at the carboxyl terminus to generate nervonic acid (24:1Δ15). This elongation reaction requires malonyl-CoA as a two-carbon donor and relies on reduced coenzyme II (NADPH) for reducing power.
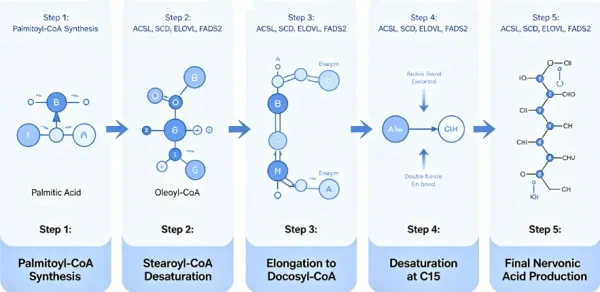
The above metabolic pathway indicates that the biosynthesis of nervonic acid directly depends on upstream fatty acids such as oleic acid, eicosenoic acid, and erucic acid. If the supply of oleic acid in the organism is insufficient, or if the activity of fatty acid elongation enzymes is inhibited, the synthesis flux of nervonic acid will decrease significantly.
In addition, some organisms (such as certain plants and microorganisms) have alternative synthetic pathways, namely the generation of nervonic acid through the elongation of erucic acid; however, this pathway has low activity in mammalian tissues.
Function of Nervonic Acid in Fatty Acid
Nervonic acid is not ubiquitous in all fatty acid metabolic pools, but rather exhibits highly specialized tissue distribution and functional orientation. In mammals, nervonic acid is mainly concentrated in the white matter region of the central nervous system, specifically within the sphingomyelin molecules of the myelin sheath.
Sphingomyelin consists of a sphingosine backbone, a phosphocholine head, and a fatty acid tail. Nervonic acid is one of the key components constituting the fatty acid tail of sphingomyelin. Its ultra-long carbon chain (24 carbons) endows sphingomyelin molecules with the following properties:
● Increasing the thickness of the lipid bilayer is beneficial for the insulating function of the myelin sheath.
● Improving the stability of lipid raft structures facilitates the anchoring of proteins related to nerve signal transduction.
● Regulating the bending stiffness of the membrane to adapt to the geometry of the axon.
From a metabolic network perspective, nervonic acid has both competitive and synergistic relationships with other fatty acids. For example, during myelin synthesis, C24:1 (nervonic acid) and C24:0 (ligninic acid, a saturated ultra-long-chain fatty acid) jointly participate in the construction of sphingomyelin, and their ratio affects the fluidity of the myelin sheath. Furthermore, nervonic acid and docosahexaenoic acid (DHA, 22:6ω-3) have different distributions in neuronal membranes: nervonic acid is mainly located in the myelin sheath, while DHA is mainly located in the synaptic membrane and the extracellular segment of rod cells.
Nervonic Acid and Other Fatty Acids in Disease
Several inherited fatty acid metabolism disorders have revealed clinically significant quantitative relationships between nervonic acid and other fatty acids. For example, in adrenoleukodystrophy (ALD), patients exhibit abnormal accumulation of saturated long-chain fatty acids (especially C24:0 and C26:0), while nervonic acid levels are relatively decreased. In clinical diagnosis, the plasma C24:0/C24:1 ratio and C26:0/C22:0 ratio are standardized biomarkers for ALD.
In the brain tissue of patients with multiple sclerosis, nervonic acid levels are 30%-50% lower than in healthy controls, while the levels of other long-chain monounsaturated fatty acids (such as oleic acid) show no significant changes. This indicates that nervonic acid has high specificity for the pathological process of demyelination.
From a supplemental therapy perspective, the intake of exogenous nervonic acid can competitively inhibit fatty acid elongation enzymes, thereby reducing the synthesis of endogenous saturated long-chain fatty acids. This mechanism has been validated in some animal models of neurodegenerative diseases, but the exact dose-response relationship in humans still requires further clinical research to confirm.
How To Choose High-quality Nervonic Acid?
Based on the aforementioned relationship between nervonic acid and fatty acids, B2B clients should pay attention to the following technical parameters when selecting nervonic acid raw materials:
• Purity Definition:
Confirm whether the purity value provided by the supplier refers to the percentage of nervonic acid methyl esters in the total fatty acid methyl esters (FAME purity) or the percentage of free nervonic acid in the total lipid extract. The difference between these two values can be 10%–20%.
• Associated Fatty Acid Profile:
Request that the supplier provide a complete quantitative list of fatty acids other than nervonic acid, with particular focus on the content of C22:1 (erucic acid), C24:0 (lignoceric acid), and C18:2 (linoleic acid).
• Oxidative Stability:
Request that the supplier provide three indicators-peroxide value, acid value, and p-anisidine value-rather than only one of them.
• Solubility and Formulation Compatibility:
Nervonic acid, as a very-long-chain monounsaturated fatty acid, is a solid, waxy substance at room temperature with a melting point of approximately 42–43°C. This characteristic affects its dispersion behavior in oil-based or water-based formulations. Customers should request the corresponding particle size specifications from the supplier based on the intended dosage form (soft capsules, microcapsule powder, emulsions, etc.).
Conclusion:
The relationship between nervonic acid and fatty acids can be summarized at three levels. At the taxonomic level, nervonic acid is a typical member of the ultra-long-chain monounsaturated ω-9 fatty acid family. At the metabolic level, the biosynthesis of nervonic acid depends on the carbon-chain elongation of upstream fatty acids such as oleic acid and erucic acid, and its in vivo level is jointly regulated by fatty acid elongation enzyme systems and desaturation enzyme systems. At the functional level, nervonic acid, together with other ultra-long-chain fatty acids (especially saturated C24:0), synergistically constitutes the hydrophobic core of myelin sphingomyelin, and changes in its content are quantitatively correlated with various demyelinating diseases.
Based on the above biochemical principles, Guanjie Biotechnology has established a comprehensive technology system for the extraction, purification, quality control, and formulation of nervonic acid fatty acids. The company focuses on the research and development of innovative bulk nervonic acid powder products, implements full-process quality control from raw materials to finished products, and supplies products to B-end customers in more than 100 countries worldwide.
For technical specifications, samples, or customized production services for nervonic acid raw materials, please contact: info@gybiotech.com.
References
[1] Sargent, J. R., Tocher, D. R., & Bell, J. G. (2002). The lipids. In: Halver, J. E., Hardy, R. W. (Eds.), Fish Nutrition (3rd ed.). Academic Press.
[2] Guillou, H., Zadravec, D., Martin, P. G. P., & Jacobsson, A. (2010). The key roles of elongases and desaturases in mammalian fatty acid metabolism. Progress in Lipid Research, 49(2), 186–199.
[3] Leonard, A. E., Pereira, S. L., Sprecher, H., & Huang, Y. S. (2004). Elongation of long-chain fatty acids. Progress in Lipid Research, 43(1), 36–54.
[4] Sandhoff, R., & Brugger, B. (2015). Sphingolipid metabolism and functions in the nervous system. FEBS Letters, 589(22), 3779–3791.
[5] O'Brien, J. S., & Sampson, E. L. (1965). Fatty acid and fatty aldehyde composition of the major brain lipids in normal human gray matter, white matter, and myelin. Journal of Lipid Research, 6(4), 537–544.
[6] Kemp, S., Berger, J., & Aubourg, P. (2012). X-linked adrenoleukodystrophy: clinical, metabolic, genetic and pathophysiological aspects. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease, 1822(9), 1465–1474.
[7] Martínez, M. (1992). Abnormal profiles of very long chain fatty acids in demyelinating diseases. Neurology, 42(4), 801–806.
[8]Jump, D. B. (2002). The biochemistry of n-3 polyunsaturated fatty acids. Journal of Biological Chemistry, 277(11), 8755–8758.










